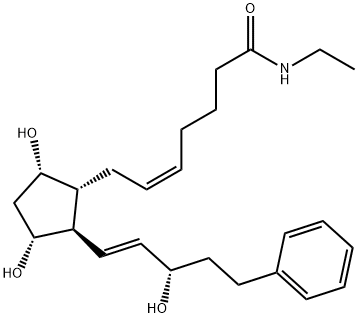
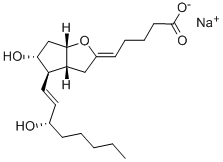
Bimatoprost , ≥99% , 155206-00-1
Synonym(s):
Bimatoprost
CAS NO.:155206-00-1
Empirical Formula: C25H37NO4
Molecular Weight: 415.57
MDL number: MFCD03411999
EINECS: 642-890-9
| Pack Size | Price | Stock | Quantity |
| 10MG | RMB119.20 | In Stock |
|
| 50MG | RMB476.80 | In Stock |
|
| 100MG | RMB788.00 | In Stock |
|
| 500MG | RMB2228.80 | In Stock |
|
| 1g | RMB3510.40 | In Stock |
|
| others | Enquire |
PRODUCT Properties
| Melting point: | 66-68°C |
| Boiling point: | 629.8±55.0 °C(Predicted) |
| Density | 1.145±0.06 g/cm3(Predicted) |
| storage temp. | -20°C |
| solubility | Chloroform (Slightly), Dichloromethane (Slightly), Methanol (Slightly) |
| form | solid |
| pka | 14.25±0.20(Predicted) |
| color | White to Off-White |
| BCS Class | 3 |
| Stability: | Light Sensitive |
| Cosmetics Ingredients Functions | EYELASH CONDITIONING |
| InChIKey | AQOKCDNYWBIDND-FTOWTWDKSA-N |
| SMILES | C(NCC)(=O)CCC/C=C\C[C@H]1[C@@H](O)C[C@@H](O)[C@@H]1/C=C/[C@@H](O)CCC1=CC=CC=C1 |
| CAS DataBase Reference | 155206-00-1(CAS DataBase Reference) |
Description and Uses
Bimatoprost was first introduced in the US and Brazil as Lumigan, an ophthalmic solution (0.03%) for the reduction of elevated intraocular pressure (IOP) in patients with open-angle glaucoma or ocular hypertension, a proven risk of glaucomatous visual field loss. Bitamoprost is a new agent belonging to the PGF2α analog class of prostamides launched in this indication after latanoprost, the most efficaceous topical medication currently available. This synthetic fatty acid amide can be prepared by esterification of 17- phenyl-18,19,20-trinorprostaglandin F2α followed by ethylamidation. Bimatoprost, as related prostamides, is devoid of typical activities associated with PGF2,, analogs; it exhibits a unique pharmacological profile in contracting the feline iris sphincter with an EC50 of 34 nM without interacting with any known prostanoid receptors. Thus it mimics the action of endogenous prostamides on specific receptors that lower IOP by increasing aqueous humor outflow through both pressure-insensitive and pressure-sensitive pathways without reducing humor formation. In a 3-month controlled clinical trial of efficacy and safety in patients with elevated IOP, bimatoprost demonstrated lower mean intraocular pressures at every time point throughout the study, as well as a good tolerance and systemic safety profile compared to latanoprost.
17-phenyl trinor Prostaglandin F2α ethyl amide (17-phenyl trinor PGF2α ethyl amide) is an F-series PG analog which has been approved for use as an ocular hypotensive drug. Investigations in our lab have shown that 17-phenyl trinor PGF2α ethyl amide is converted by an amidase enzymatic activity in the bovine and human cornea to yield the corresponding free acid, with a conversion rate of about 40 μg/g corneal tissue/24 hours. The free acid, 17-phenyl trinor PGF2α, is a potent FP receptor agonist. In human and animal models of glaucoma, FP receptor agonist activity corresponds very closely with intraocular hypotensive activity.[Cayman Chemical]
Safety
| Symbol(GHS) |   GHS07,GHS08 |
| Signal word | Danger |
| Hazard statements | H302-H360F |
| Precautionary statements | P201-P301+P312+P330-P308+P313 |
| PPE | Eyeshields, Gloves, type N95 (US) |
| WGK Germany | 3 |
| Storage Class | 6.1C - Combustible acute toxic Cat.3 toxic compounds or compounds which causing chronic effects |
| Hazard Classifications | Acute Tox. 4 Oral Repr. 1B |