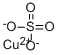A6353730
Cupric Sulfate CS
Synonym(s):
Cupric sulfate;Copper(II) sulfate;Copper(II) sulfate solution;Cupric sulfate solution acc. to Fehling;Cupric sulfate standard
CAS NO.:
Empirical Formula: CuO4S
Molecular Weight: 159.61
MDL number: MFCD00010981
EINECS: 231-847-6
| Pack Size | Price | Stock | Quantity |
| 100ml | RMB1039.20 | In Stock |
|
| others | Enquire |
Update time: 2022-07-08
PRODUCT Properties
| Melting point: | 200 °C (dec.)(lit.) |
| bulk density | 800kg/m3 |
| Density | 3.603 g/mL at 25 °C(lit.) |
| vapor pressure | 7.3 mm Hg ( 25 °C) |
| storage temp. | Store at +5°C to +30°C. |
| solubility | H2O: soluble |
| form | powder |
| color | Slightly greenish to gray |
| Specific Gravity | 3.603 |
| Flame Color | Bluish-green |
| Odor | odorless |
| PH | 3.5-4.5 (50g/l, H2O, 20℃) |
| PH Range | 3.7 - 4.5 |
| Water Solubility | 203 g/L (20 ºC) |
| Thermal Conductivity | 2.092 W/(m·K) (crystal) |
| Sensitive | Hygroscopic |
| Merck | 14,2653 |
| Exposure limits | ACGIH: TWA 1 mg/m3 NIOSH: IDLH 100 mg/m3; TWA 1 mg/m3 |
| Dielectric constant | 10.3(0.0℃) |
| Stability: | hygroscopic |
| Cosmetics Ingredients Functions | SKIN CONDITIONING |
| InChI | 1S/Cu.H2O4S/c;1-5(2,3)4/h;(H2,1,2,3,4)/q+2;/p-2 |
| InChIKey | ARUVKPQLZAKDPS-UHFFFAOYSA-L |
| SMILES | [Cu++].[O-]S([O-])(=O)=O |
| CAS DataBase Reference | 7758-98-7(CAS DataBase Reference) |
| NIST Chemistry Reference | Cupric sulfate(7758-98-7) |
| EPA Substance Registry System | Cupric sulfate (7758-98-7) |
Description and Uses
Copper sulfate is a greenish-white crystallinesolid; the pentahydrate is blue powder or granules, or ultramarine crystalline solid. Molecular weight =159.60; 249.70(pentahydrate). Boiling point =150℃ (pentahydrate) with25H2O; Melting point = (decomposes) .200℃; decomposes .110℃ (pentahydrate) with 24H2O; copper sulfatedecomposes to CuO at 650℃. Hazard Identification (basedon NFPA-704 M Rating System): Health 2, Flammability 0,Reactivity 0. Highly soluble in water; forms a bright bluesolution.
Copper(II) sulfate may be employed for the following studies:
- As a catalyst for the acetylation of alcohols and phenols under solvent-free conditions.
- To compose the electrolyte for the electrodeposition of Cu-Zn-Sn precursors, required for the preparation of Cu2ZnSnS4 (CZTS) thin films.
- As a Lewis acid catalyst for the dehydration of alcohols.5
Safety
| Symbol(GHS) |   GHS05,GHS09 |
| Signal word | Danger |
| Hazard statements | H318-H410 |
| Precautionary statements | P273-P280-P305+P351+P338-P391-P501 |
| PPE | dust mask type N95 (US), Eyeshields, Faceshields, Gloves |
| Hazard Codes | Xn,N,Xi |
| Risk Statements | 36/38-50/53-22-51/53-36/37/38 |
| Safety Statements | 24/25-36-60-61-22-26 |
| RIDADR | UN 3288 6.1/PG 3 |
| WGK Germany | 2 |
| RTECS | GL8800000 |
| F | 3 |
| TSCA | TSCA listed |
| HS Code | 2833 25 00 |
| HazardClass | 6.1 |
| PackingGroup | III |
| Storage Class | 11 - Combustible Solids |
| Hazard Classifications | Acute Tox. 4 Oral Aquatic Acute 1 Aquatic Chronic 1 Eye Irrit. 2 Skin Irrit. 2 |
| Hazardous Substances Data | 7758-98-7(Hazardous Substances Data) |
| Toxicity | LD50 orally in Rabbit: 481 mg/kg |
| Limited Quantities | 5.0 L (1.3 gallons) (liquid) or 5.0 kg (11 lbs) (solid) |
| Excepted Quantities | Max Inner Pack (30g or 30ml) and Max Outer Pack (1Kg or 1L) |