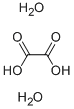
Oxalic acid dihydrate , StandardforGC,≥99.6% , 6153-56-6
Synonym(s):
Ethanedioic acid;Ethanedioic acid dihydrate;Ethanedioic Acid, Dihydrate, Ethanedioic Acid;Oxalic acid dihydrate;Oxalic Acid, Dihydrate
CAS NO.:6153-56-6
Empirical Formula: C2H6O6
Molecular Weight: 126.07
MDL number: MFCD00149102
EINECS: 612-167-2
| Pack Size | Price | Stock | Quantity |
| 5G | RMB51.20 | In Stock |
|
| others | Enquire |
PRODUCT Properties
| Melting point: | 104-106 °C(lit.) |
| Boiling point: | 108-109°C |
| bulk density | 813kg/m3 |
| Density | 1,65 g/cm3 |
| vapor density | 4.4 (vs air) |
| vapor pressure | <0.01 mm Hg ( 20 °C) |
| Flash point: | 157°C |
| storage temp. | Store at +5°C to +30°C. |
| solubility | H2O: soluble1M at 20°C, clear, colorless |
| form | Powder/Solid |
| color | Yellow to yellow-green |
| Specific Gravity | 1.65 |
| PH | ~1.0 (25℃, 1M in H2O) |
| PH Range | 6 - 8 at 25 °C |
| Water Solubility | 138 g/L (20 ºC) |
| Sublimation | 157 ºC |
| Merck | 14,6911 |
| BRN | 3679436 |
| Exposure limits | TLV-TWA for anhydrous acid 1 mg/m3
(ACGIH, MSHA, and OSHA); TLV-STEL
2 mg/m3 (ACGIH). |
| Stability: | Stable. Incompatible with bases, acid chlorides, steel, silver, silver compounds, moisture. Avoid contact with metals. |
| InChI | 1S/C2H2O4.2H2O/c3-1(4)2(5)6;;/h(H,3,4)(H,5,6);2*1H2 |
| InChIKey | GEVPUGOOGXGPIO-UHFFFAOYSA-N |
| SMILES | [H]O[H].[H]O[H].OC(=O)C(O)=O |
| CAS DataBase Reference | 6153-56-6(CAS DataBase Reference) |
| NIST Chemistry Reference | Oxalic acid dihydrate(6153-56-6) |
| EPA Substance Registry System | Oxalic acid dihydrate (6153-56-6) |
| Absorption | cut-off at 303nm in H2O at 1M |
Description and Uses
Oxalic acid occurs in the cell sap of Oxalisand Rumex species of plants as the potassium and calcium salt. It is the metabolicproduct of many molds (Merck 1989). Thereare a large number of applications of thiscompound, including indigo dyeing; calicoprinting; removal of paint, rust, and inkstains; metal polishing; bleaching leather; inpesticide compositions and manufacture ofoxalates. It is also used as an analyticalreagent and as a reducing agent in organicsynthesis.
Addition of oxalic acid to chromic acid forthe anodizing of Al alloy has been reported tomodify the morphology and improve the corrosion performance of anodic films (Moutarlier et al. 2004). Also, it is a very effectiveadditive for the ozone treatment of cellulose.It prevents the degradation of cellulose fromozone bleaching.
Safety
| Symbol(GHS) |   GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | H302+H312-H318 |
| Precautionary statements | P264-P270-P280-P301+P312-P302+P352+P312-P305+P351+P338 |
| PPE | dust mask type N95 (US), Eyeshields, Gloves |
| Hazard Codes | Xn,C |
| Risk Statements | 21/22-41 |
| Safety Statements | 24/25-39-37-36-26 |
| RIDADR | UN 3261 8/PG 3 |
| WGK Germany | 1 |
| RTECS | RO2450000 |
| TSCA | Yes |
| HazardClass | 8 |
| PackingGroup | III |
| HS Code | 29171100 |
| Storage Class | 11 - Combustible Solids |
| Hazard Classifications | Acute Tox. 4 Dermal Acute Tox. 4 Oral Eye Dam. 1 |
| Toxicity | LD50 orally in Rabbit: 375 mg/kg |