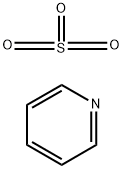
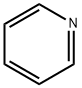
Pyridine , AR,≥99.5% , 110-86-1
Synonym(s):
Azabenzene;Azine, Azabenzene;Pyridine
CAS NO.:110-86-1
Empirical Formula: C5H5N
Molecular Weight: 79.1
MDL number: MFCD00011732
EINECS: 203-809-9
PRODUCT Properties
| Melting point: | -42 °C (lit.) |
| Boiling point: | 115 °C (lit.) |
| Density | 0.978 g/mL at 25 °C (lit.) |
| vapor density | 2.72 (vs air) |
| vapor pressure | 23.8 mm Hg ( 25 °C) |
| refractive index | n |
| FEMA | 2966 | PYRIDINE |
| Flash point: | 68 °F |
| storage temp. | Store at +5°C to +30°C. |
| solubility | H2O: in accordance |
| pka | 5.25(at 25℃) |
| form | Liquid |
| color | colorless |
| PH | 8.81 (H2O, 20℃) |
| Relative polarity | 0.302 |
| Odor | Nauseating odor detectable at 0.23 to 1.9 ppm (mean = 0.66 ppm) |
| Odor Threshold | 0.063ppm |
| Odor Type | fishy |
| biological source | synthetic |
| explosive limit | 12.4% |
| Water Solubility | Miscible |
| FreezingPoint | -42℃ |
| Thermal Conductivity | 0.162 W/(m·K) at 25 ℃ |
| λmax | λ: 305 nm Amax: 1.00 λ: 315 nm Amax: 0.15 λ: 335 nm Amax: 0.02 λ: 350-400 nm Amax: 0.01 |
| Merck | 14,7970 |
| BRN | 103233 |
| Henry's Law Constant | 18.4 at 30 °C (headspace-GC, Chaintreau et al., 1995) |
| Exposure limits | TLV-TWA 5 ppm (~15 mg/m3) (ACGIH,
MSHA,and OSHA); STEL 10 ppm (ACGIH),
IDLH 3600 ppm (NIOSH). |
| Dielectric constant | 12.5(20℃) |
| Stability: | Stable. Flammable. Incompatible with strong oxidizing agents, strong acids. |
| InChI | 1S/C5H5N/c1-2-4-6-5-3-1/h1-5H |
| InChIKey | JUJWROOIHBZHMG-UHFFFAOYSA-N |
| SMILES | C1=CN=CC=C1 |
| LogP | 0.64 at 20℃ |
| Surface tension | 38mN/m at 20°C |
| CAS DataBase Reference | 110-86-1(CAS DataBase Reference) |
| IARC | 2B (Vol. 77, 119) 2019 |
| NIST Chemistry Reference | Pyridine(110-86-1) |
| EPA Substance Registry System | Pyridine (110-86-1) |
Description and Uses
Pyridine is used directly in the denaturation of alcohol (ACGIH 1986; HSDB 1989; NSC 1978) and as a solvent in paint and rubber preparation (ACGIH 1986; HSDB 1989; NSC 1978) and in research laboratories for functions such as extracting plant hormones (Santodonato et al. 1985). Half of the pyridine produced today is used as an intermediate in making various insecticides and herbicides for agricultural applications (ACGIH 1986; Harper et al. 1985; Santodonato et al. 1985). Approximately 20% goes into the production of piperidine (Harper et al. 1985; Santodonato et al. 1985) which is commercially significant in the preparation of chemicals used in rubber vulcanization and agriculture (NSC 1978). Pyridine is also used as an intermediate in the preparation of drugs (antihistamines, steroids, sulfa-type and other antibacterial agents) dyes, water repellents, and polycarbonate resins (ACGIH 1986; Harper et al. 1985; NSC 1978; Santodonato et al. 1985). Pyridine is also approved by the Food and Drug Administration (FDA) for use as a flavoring agent in the preparation of foods (Harper et al. 1985; HSDB 1989) .
Safety
| Symbol(GHS) |   GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H225-H302+H312+H332-H315-H319 |
| Precautionary statements | P210-P280-P301+P312-P303+P361+P353-P304+P340+P312-P305+P351+P338 |
| Hazard Codes | T,N,F,Xn |
| Risk Statements | 11-20/21/22-39/23/24/25-23/24/25-52-36/38 |
| Safety Statements | 36/37/39-38-45-61-28A-26-28-24/25-22-36/37-16-7 |
| RIDADR | UN 1282 |
| OEB | A |
| OEL | TWA: 5 ppm (15 mg/m3) |
| WGK Germany | 2 |
| RTECS | UR8400000 |
| F | 3-10 |
| Autoignition Temperature | 482 °C |
| Hazard Note | Highly Flammable/Harmful |
| TSCA | TSCA listed |
| HS Code | 2933 31 00 |
| HazardClass | 3 |
| PackingGroup | II |
| Storage Class | 3 - Flammable liquids |
| Hazard Classifications | Acute Tox. 4 Dermal Acute Tox. 4 Inhalation Acute Tox. 4 Oral Eye Irrit. 2 Flam. Liq. 2 Skin Irrit. 2 |
| Hazardous Substances Data | 110-86-1(Hazardous Substances Data) |
| Toxicity | LD50 orally in rats: 1.58 g/kg (Smyth) |
| IDLA | 1,000 ppm |
| Limited Quantities | 1.0 L (0.3 gallon) (liquid) |
| Excepted Quantities | Max Inner Pack (30g or 30ml) and Max Outer Pack (500g or 500ml) |

![1,2,4-Triazolo[4,3-<i>a</i>]pyridin-3(2<i>H</i>)-one](https://img.chemicalbook.com/CAS/GIF/6969-71-7.gif)