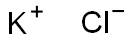Potassium chloride concentrate , 0.1M , 7447-40-7
Synonym(s):
Potassium chloride;KCl;Potassium chloride solution;CLK;CLK1
CAS NO.:7447-40-7
Empirical Formula: ClK
Molecular Weight: 74.55
MDL number: MFCD00011360
EINECS: 231-211-8
| Pack Size | Price | Stock | Quantity |
| 50ML | RMB167.20 | In Stock |
|
| others | Enquire |
PRODUCT Properties
| Melting point: | 770 °C (lit.) |
||||||||||||||
| Boiling point: | 1420°C |
||||||||||||||
| bulk density | 1000kg/m3 |
||||||||||||||
| Density | 1.98 g/mL at 25 °C (lit.) |
||||||||||||||
| refractive index | n |
||||||||||||||
| Flash point: | 1500°C |
||||||||||||||
| storage temp. | 2-8°C |
||||||||||||||
| solubility | H2O: soluble |
||||||||||||||
| form | random crystals |
||||||||||||||
| color | White |
||||||||||||||
| Specific Gravity | 1.984 |
||||||||||||||
| Odor | Odorless |
||||||||||||||
| PH | 5.5-8.0 (20℃, 50mg/mL in H2O) |
||||||||||||||
| PH Range | 7 |
||||||||||||||
| Flame Color | Light Purple |
||||||||||||||
| biological source | rabbit |
||||||||||||||
| Water Solubility | 340 g/L (20 ºC) |
||||||||||||||
| Thermal Conductivity | 8.368 W/(m·K) |
||||||||||||||
| λmax | λ: 260 nm Amax: 0.02 λ: 280 nm Amax: 0.01 |
||||||||||||||
| Sensitive | Hygroscopic |
||||||||||||||
| Crystal Structure | NaCl type |
||||||||||||||
| crystal system | Cube |
||||||||||||||
| Merck | 14,7621 |
||||||||||||||
| Sublimation | 1500 ºC |
||||||||||||||
| Space group | Fm3m |
||||||||||||||
| BRN | 1711999 |
||||||||||||||
| Lattice constant |
|
||||||||||||||
| Dielectric constant | 4.6(Ambient) |
||||||||||||||
| BCS Class | 1 |
||||||||||||||
| Stability: | Stable. Incompatible with strong oxidizing agents, strong acids. Protect from moisture. Hygroscopic. |
||||||||||||||
| Cosmetics Ingredients Functions | VISCOSITY CONTROLLING |
||||||||||||||
| InChI | 1S/ClH.K/h1H;/q;+1/p-1 |
||||||||||||||
| InChIKey | WCUXLLCKKVVCTQ-UHFFFAOYSA-M |
||||||||||||||
| SMILES | [Cl-].[K+] |
||||||||||||||
| CAS DataBase Reference | 7447-40-7(CAS DataBase Reference) |
||||||||||||||
| NIST Chemistry Reference | Potassium chloride(7447-40-7) |
||||||||||||||
| EPA Substance Registry System | Potassium chloride (7447-40-7) |
Description and Uses
Potassium chloride (KCl) is a metal halide salt that is used in a variety of areas. The dominant application of potassium chloride is to serve as a fertilizer, which offers potassium to plants and prevents them from certain diseases. Besides, it can be applied in food and medical industry. As a treatment for hypokalemia, potassium chloride pills are taken to balance the blood's potassium levels and prevent potassium deficiency in the blood. In food industry, it serves as a electrolyte replenisher and a good salt substitute for food, as well as a firming agent to give consistent texture to food, thus to strengthen its structure.
About 4-5% of potash production is used in industrial applications (UNIDOIFDC, 1998). In 1996, the world supply of industrial grade potash was close to 1.35 Mt K2O. This industrial material is 98-99% pure, compared with the agricultural potash specification of 60% K2O minimum (equivalent to 95% KCl). Industrial potash should contain at least 62% K2O and have very low levels of Na, Mg, Ca, SO4 and Br. This high-grade potash is produced by only a few producers in worldwide.
Potassium hydroxide (KOH), also known as caustic potash, is the largestvolume K product for non-fertilizer use. It is produced by the electrolysis of industrial KCl and is widely used for manufacturing soaps, detergents, grease, catalysts, synthetic rubber, matches, dyes and insecticides. Caustic potash is also as a liquid fertilizer and as an ingredient in alkaline batteries and photographic film processing chemicals.
Potassium hydroxide is a raw material in the production of various K salts, mainly K carbonates, and also citrates, silicates, acetates, etc. Potassium carbonate confers excellent clarity to glass thus is used for most fine optical lenses, eyeglasses, fine crystal, glassware, chinaware and TV tubes. Potassium bicarbonate is used largely in the food and pharmaceutical industries.
Potash-derived compounds and salts are also used in the production of metal fluxes, cured meats, tempered steel, paper fumigants, case hardened steel, bleaching agents, baking powder, cream of tartar and beverages. Worldwide, industrial KCl is estimated to be used as follows: detergents and soaps, 30-35%; glass and ceramics, 25-28%; textiles and dyes 20-22%; chemicals and drugs, 13-15%; and other uses, 7-5% (UNIDO-IFDC, 1998).
Safety
| Symbol(GHS) |  GHS08 |
| Signal word | Danger |
| Hazard statements | H303 |
| Precautionary statements | P201-P308+P313 |
| Hazard Codes | Xi,C,F,Xn,T |
| Risk Statements | 36-34-11-36/37/38-40-61-60 |
| Safety Statements | 24/25-39-26-22-23-45-36/37/39-16-36/37-53 |
| RIDADR | UN 1824 8 / PGII |
| WGK Germany | 1 |
| RTECS | TS8050000 |
| F | 8 |
| TSCA | TSCA listed |
| HS Code | 31042090 |
| Storage Class | 13 - Non Combustible Solids |
| Hazardous Substances Data | 7447-40-7(Hazardous Substances Data) |
| Toxicity | LD50 orally in Rabbit: 2600 mg/kg |