Tamibarotene, a selective agonist of the retinoic acid receptor, was launched in
Japan as an oral treatment for relapsed or refractory acute promyelocytic
leukemia (APL). APL is a form of acute myeloid leukemia (AML) characterized by
a deficiency in mature blood cells and an excess of immature cells called promyelocytes
in the bone marrow and peripheral blood. The current standard of care
for APL includes treatment with all-trans-retinoic acid (ATRA), either alone or in
combination with chemotherapy. ATRA is a high affinity ligand for two types of
nuclear receptors, retinoic acid receptor (RAR) and retinoid X receptor (RXR),
each of which has three subtypes (-α, -β, and -γ). Activation of RARα by ATRA
causes promyelocytes to differentiate and mature, thereby inhibiting their proliferation
and inducing disease remission. Although ATRA is one of the most clinically
successful retinoids, its usage is hampered by the high rate of adverse effects,
instability, and the appearance of ATRA-resistant leukemia cells.Adverse events included retinoic acid syndrome, hyperleukocytosis, xerosis,
cheilitis, hypertriglyceridemia, and hypercholesterolemia; however, these side effects
were generally milder than with ATRA, which all patients had received previously.
Examination of human samples taken from Phase II and III clinical trials revealed
that fecal excretion was the major elimination route, and the metabolism of tamibarotene
occurred primarily through hydroxylation and taurine conjugation. In
vitro, the plasma protein binding of tamibarotene is shown to be >98% in rats,
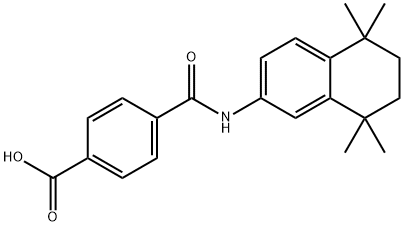
dogs, and humans. Tamibarotene is synthesized from 5,5,8,8-tetramethyl-5,6,7,
8-tetrahydronaphthalene in a four-step sequence consisting of regioselective nitration
in the 2-position, reduction of the nitro group by hydrogenation to produce
the corresponding aniline derivative, acylation of the aniline intermediate with
4-(carbomethoxy)benzoyl chloride, and hydrolysis of the methyl ester.
Tamibarotene, a retinoic acid receptor-α(RARα) agonist,
was approved for the treatment of relapsed or refractory
acute promyelocytic leukemia (APL) in Japan on June, 2005
and is currently marketed by Nippon Shinyaku Co. This
novel drug has shown high remission rate among patients
who have recurrent disease after all trans retinoic acid therapy.