Urea , 99.999%metalsbasis , 57-13-6
Synonym(s):
Urea;Carbamide;NBK;Urea solution;Ureum
CAS NO.:57-13-6
Empirical Formula: CH4N2O
Molecular Weight: 60.06
MDL number: MFCD00008022
EINECS: 200-315-5
| Pack Size | Price | Stock | Quantity |
| 25G | RMB70.40 | In Stock |
|
| 100G | RMB237.60 | In Stock |
|
| 500G | RMB952.00 | In Stock |
|
| 5KG | RMB5712.00 | In Stock |
|
| others | Enquire |
PRODUCT Properties
| Melting point: | 132-135 °C(lit.) |
| Boiling point: | 332.48°C (estimate) |
| bulk density | 720-760kg/m3 |
| Density | 1.335 g/mL at 25 °C(lit.) |
| vapor pressure | <0.1 hPa (20 °C) |
| refractive index | n |
| storage temp. | 2-8°C |
| solubility | H2O: 8 M at 20 °C |
| pka | 0.10(at 25℃) |
| form | powder |
| color | white |
| Specific Gravity | 1.335 |
| Odor | almost odorless |
| PH | 8.0-10.0 (20℃, 8M in H2O) |
| biological source | synthetic |
| Water Solubility | 1080 g/L (20 ºC) |
| λmax | λ: 260 nm Amax: 0.03 λ: 280 nm Amax: 0.02 |
| Merck | 14,9867 |
| BRN | 635724 |
| Henry's Law Constant | 5.7×106 mol/(m3Pa) at 25℃, Duchowicz et al. (2020) |
| Dielectric constant | 3.5(Ambient) |
| Stability: | Substances to be avoided include strong oxidizing agents. Protect from moisture. |
| Cosmetics Ingredients Functions | BUFFERING HUMECTANT SKIN CONDITIONING ANTISTATIC |
| InChI | 1S/CH4N2O/c2-1(3)4/h(H4,2,3,4) |
| InChIKey | XSQUKJJJFZCRTK-UHFFFAOYSA-N |
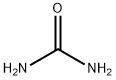
| SMILES | NC(N)=O |
| LogP | -1.660 (est) |
| CAS DataBase Reference | 57-13-6(CAS DataBase Reference) |
| NIST Chemistry Reference | Urea(57-13-6) |
| EPA Substance Registry System | Urea (57-13-6) |
| Absorption | ≤0.06 at 260nm at 5M ≤0.06 at 280nm at 5M |
Description and Uses
Urea is a stable highly water-soluble compound
of high nitrogen content (47%), with good storage
properties that make it the most commonly used nitrogen
fertilizer. The synthesis process has remained essentially
unchanged since it was first developed by the BASF
Corporation in 1922. In this process, liquid ammonia
is reacted with carbon dioxide to produce ammonium
carbamate, which is then dehydrated to form urea. The
reactions are:
2NH3 + CO2 === NH2·CO2·NH4
NH2·CO2·NH4 === (NH2)2CO + H2O
Urea is a physiological regulator of nitrogen excretion in mammals; synthesized in the liver as an end-product of protein catabolism and excreted in urine. Also occurs normally in skin. Emollient; diu retic.
Safety
| Symbol(GHS) |  GHS07 |
| Signal word | Warning |
| Hazard statements | H320 |
| Precautionary statements | P264-P337+P313-P305+P351+P338 |
| PPE | Eyeshields, Gloves, type N95 (US) |
| Hazard Codes | Xn,Xi |
| Risk Statements | 36/37/38-40-38 |
| Safety Statements | 26-36-24/25-37 |
| RIDADR | Not regulated |
| WGK Germany | 1 |
| RTECS | YR6250000 |
| TSCA | TSCA listed |
| HS Code | 31021010 |
| Storage Class | 11 - Combustible Solids |
| Hazardous Substances Data | 57-13-6(Hazardous Substances Data) |
| Toxicity | LD50 orally in Rabbit: 8471 mg/kg LD50 dermal Rat 8200 mg/kg |