A1680512
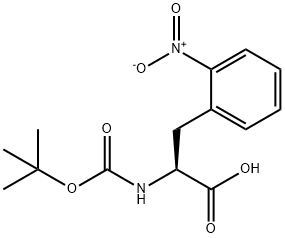
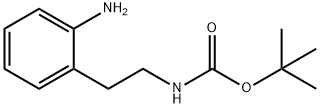
Boc-2-nitro-<SC>L</SC>-phenylalanine , 98%(HPLC) , 185146-84-3
| Pack Size | Price | Stock | Quantity |
| 250mg | RMB117.60 | In Stock |
|
| 1G | RMB296.00 | In Stock |
|
| 5g | RMB1480.00 | In Stock |
|
| 10g | RMB2837.60 | In Stock |
|
| others | Enquire |
Update time: 2022-07-08
PRODUCT Properties
| Boiling point: | 488.0±40.0 °C(Predicted) |
| Density | 1.290±0.06 g/cm3(Predicted) |
| storage temp. | 2-8°C |
| form | solid |
| pka | 3.69±0.11(Predicted) |
| Major Application | peptide synthesis |
| InChI | 1S/C14H18N2O6/c1-14(2,3)22-13(19)15-10(12(17)18)8-9-6-4-5-7-11(9)16(20)21/h4-7,10H,8H2,1-3H3,(H,15,19)(H,17,18)/t10-/m0/s1 |
| InChIKey | BSHMNGMAJNWNBP-JTQLQIEISA-N |
| SMILES | OC([C@@H](NC(OC(C)(C)C)=O)CC1=C([N+]([O-])=O)C=CC=C1)=O |
| CAS DataBase Reference | 185146-84-3(CAS DataBase Reference) |
Description and Uses
Boc-L-2-Nitrophenylalanine is a protected derivative of L-Phenylalanine (P319415), and is used in the photocleavage of polypeptide backbones. L-Phenylalanine is an essential amino acid. L-Phenylalanine is biologically converted into L-tyrosine, another one of the DNA-encoded amino acids, which in turn is converted to L-DOPA and further converted into dopamine, norepinephrine, and epinephrine.
Safety
| Hazard Codes | Xi |
| WGK Germany | WGK 3 |
| HazardClass | IRRITANT |
| HS Code | 2922498590 |
| Storage Class | 11 - Combustible Solids |