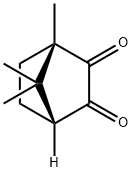
A7158712
(1<i>R</i>)-(-)-Camphorquinone , >98.0%(GC) , 10334-26-6
Synonym(s):
(1R)-(−)-2,3-Bornanedione;2,3-Bornanedione
CAS NO.:10334-26-6
Empirical Formula: C10H14O2
Molecular Weight: 166.22
MDL number: MFCD00082863
EINECS: 626-988-9
| Pack Size | Price | Stock | Quantity |
| 1G | RMB215.20 | In Stock |
|
| 5G | RMB703.20 | In Stock |
|
| others | Enquire |
Update time: 2022-07-08
PRODUCT Properties
| Melting point: | 200-203 °C(lit.) |
| alpha | -101° (20/D)(c=2, C6H5CH3) |
| Boiling point: | 254.44°C (rough estimate) |
| Density | 1.0060 (rough estimate) |
| refractive index | 1.5200 (estimate) |
| storage temp. | Sealed in dry,Room Temperature |
| solubility | almost transparency in Methanol |
| form | powder to crystal |
| color | Light yellow to Yellow |
| optical activity | [α]20/D 101°, c = 2 in toluene |
| BRN | 2327696 |
| InChI | InChI=1S/C10H14O2/c1-9(2)6-4-5-10(9,3)8(12)7(6)11/h6H,4-5H2,1-3H3/t6-,10+/m1/s1 |
| InChIKey | VNQXSTWCDUXYEZ-LDWIPMOCSA-N |
| SMILES | [C@]12(C)C(C)(C)[C@]([H])(CC1)C(=O)C2=O |
| CAS DataBase Reference | 10334-26-6(CAS DataBase Reference) |
Description and Uses
(1R)-()-Camphorquinone can be used as a chiral starting material for the preparation of:
- α-Hydroxycamphors by selective reduction of keto groups using various vegetables.
- Camphor-1,2-diamine platinum(II) complexes for DNA interaction studies.
- Camphoric anhydride by unsensitized photo-oxidation in the presence of oxygen and polar solvents.
- Camphorquinone-based chiral homoallylic amine, which is reacted with aldehydes to produce homoallylic primary amines via imine formation followed by 2-azonia-Cope rearrangement.
Safety
| Symbol(GHS) |   GHS07,GHS08 |
| Signal word | Danger |
| Hazard statements | H302-H334 |
| Precautionary statements | P301+P312+P330 |
| PPE | dust mask type N95 (US), Eyeshields, Faceshields, Gloves |
| Hazard Codes | Xn |
| Risk Statements | 22-43 |
| Safety Statements | 36/37 |
| WGK Germany | 3 |
| HS Code | 2914.29.5000 |
| Storage Class | 11 - Combustible Solids |
| Hazard Classifications | Acute Tox. 4 Oral Resp. Sens. 1 |

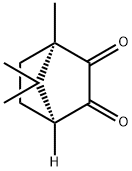
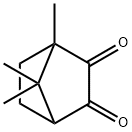
![(7,7-Dimethyl-2,3-dioxobicyclo[2.2.1]Heptan-1-yl)methanesulfonicacidhydrate](https://img.chemicalbook.com/CAS/GIF/73413-79-3.gif)