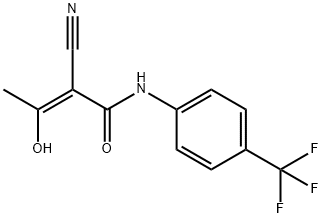
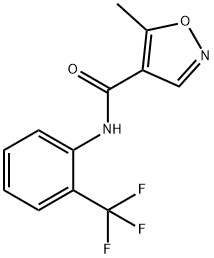
Teriflunomide , ≥99% , 163451-81-8
CAS NO.:163451-81-8
Empirical Formula: C12H9F3N2O2
Molecular Weight: 270.21
MDL number: MFCD00910058
EINECS: 642-273-4
| Pack Size | Price | Stock | Quantity |
| 50MG | RMB159.20 | In Stock |
|
| 250MG | RMB559.20 | In Stock |
|
| 1g | RMB1439.20 | In Stock |
|
| 5g | RMB3199.20 | In Stock |
|
| others | Enquire |
PRODUCT Properties
| Melting point: | 229-232°C |
| Boiling point: | 410.8±45.0 °C(Predicted) |
| Density | 1.424±0.06 g/cm3(Predicted) |
| storage temp. | -20°C |
| solubility | DMSO: soluble5mg/mL, clear (warmed) |
| pka | 5.20±0.50(Predicted) |
| form | powder |
| color | white to beige |
| λmax | 203nm(MeOH)(lit.) |
| Merck | 14,9165 |
| Stability: | Hygroscopic |
| InChI | InChI=1S/C12H9F3N2O2/c1-7(18)10(6-16)11(19)17-9-4-2-8(3-5-9)12(13,14)15/h2-5,18H,1H3,(H,17,19)/b10-7- |
| InChIKey | UTNUDOFZCWSZMS-YFHOEESVSA-N |
| SMILES | C(NC1=CC=C(C(F)(F)F)C=C1)(=O)/C(/C#N)=C(\O)/C |
Description and Uses
A-771726 is an active metabolite of the prodrug leflunomide that inhibits dihydroorotate dehydrogenase (DHODH; IC50 = 0.23 μM). A-771726 inhibits the production of prostaglandin E2 (PGE2; ) in TNF-α- or IL-1α-stimulated isolated human synoviocytes (IC50s = 7 and 3 μM, respectively). It inhibits the proliferation of isolated human peripheral blood mononuclear cells (PBMCs) when used at concentrations of 25 and 100 μM. A-771726 (3 and 10 mg/kg) delays disease onset and decreases neurological deficits in a rat model of experimental autoimmune encephalomyelitis (EAE) induced by complete Freund’s adjuvant (CFA) and M. tuberculosis. Formulations containing teriflunomide have been used in the treatment of multiple sclerosis.
A-771726 is the active metabolite of leflunomide, a prodrug approved by the FDA for treatment of rheumatoid arthritis. A-771726 reversibly inhibits dihydroorotate dehydrogenase, the rate-limiting step in the de novo synthesis of pyrimidines. It prevents activated lymphocytes from accumulating sufficient pyrimidines to support DNA synthesis (IC50s = 0.09, 3.5, and 12.5 μM for rat, mouse, and human, respectively). At higher doses, A-771726 inhibits tyrosine kinases responsible for early T cell and B cell signaling in the G0/G1 phase of the cell cycle. A-771726 has also been shown to inhibit the production of prostaglandin E2 in synoviocytes activated by TNF-α and IL-1α (IC50s = 7 and 3 μM, respectively) as well as inhibit MMP-1 and IL-6 production at concentrations >10 μM.[Cayman Chemical]
Safety
| Symbol(GHS) |  GHS07 |
| Signal word | Warning |
| Hazard statements | H225-H302+H312+H332-H319 |
| Precautionary statements | P210-P280-P301+P312-P303+P361+P353-P304+P340+P312-P305+P351+P338 |
| Hazard Codes | Xn |
| Risk Statements | 22 |
| WGK Germany | 3 |
| HS Code | 2926.90.4801 |
| Storage Class | 11 - Combustible Solids |
| Hazard Classifications | Acute Tox. 4 Oral |